|
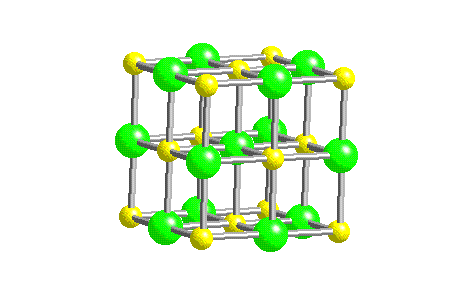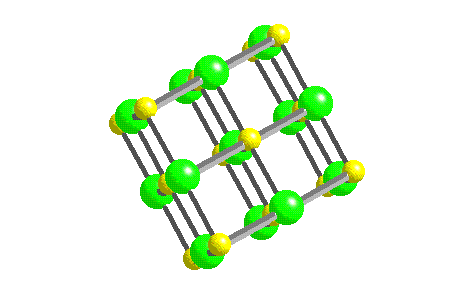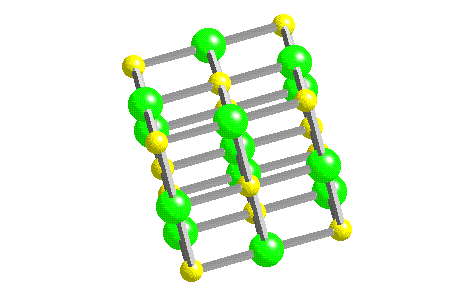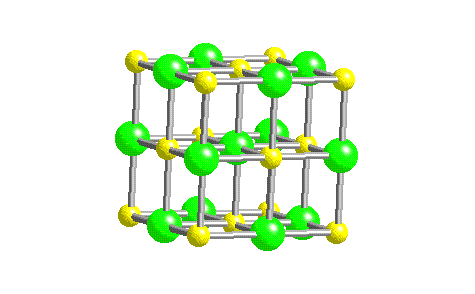
In such an arrangement each cation in the lattice is surrounded by more than one anion (typically four, six, or eight) and vice versa, so it is more stable than a system consisting of separate pairs of ions, in which there is only one cation–anion interaction in each pair. 
While Equation 4.1.1 has demonstrated that the formation of ion pairs from isolated ions releases large amounts of energy, even more energy is released when these ion pairs condense to form an ordered three-dimensional array. 
These properties result from the regular arrangement of the ions in the crystalline lattice and from the strong electrostatic attractive forces between ions with opposite charges. They are not easily deformed, and they melt at relatively high temperatures. Ionic compounds are usually rigid, brittle, crystalline substances with flat surfaces that intersect at characteristic angles. Metal ores are commonly combinations of metal atoms with oxygen atoms, and this combination is produced when metals rust, so the process where electrons are transferred to the oxygen atoms from the metal atoms is known as oxidation of the metal and the reverse process, where pure metals are produced is called reduction of the ore to the metal. The reaction of a metal with a nonmetal usually produces an ionic compound that is, electrons are transferred from the metal to the nonmetal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
